Omnipod 5: A Comprehensive Review of the Tubeless Automated Insulin Delivery System
The Omnipod 5 Automated Insulin Delivery (AID) System stands as a notable advancement in contemporary diabetes management. This tubeless, wearable solution is meticulously engineered to streamline insulin administration and enhance glycemic control. This extensive review will meticulously examine the system’s innovative functionalities, clinical efficacy, potential constraints, and authentic user experiences, thereby offering an evidence-based perspective for individuals contemplating the adoption of this sophisticated technology.
Introduction to Omnipod 5
The Omnipod 5 distinguishes itself as the inaugural tubeless AID system that seamlessly integrates with continuous glucose monitoring (CGM) to autonomously regulate insulin delivery. Developed by Insulet, this system endeavors to alleviate the daily complexities associated with diabetes management by proactively modulating insulin dosages to mitigate both hyperglycemic and hypoglycemic events. Its regulatory approval extends to individuals with type 1 diabetes from two years of age and older, and those with type 2 diabetes aged eighteen years and above.
Key Features and Technological Innovations
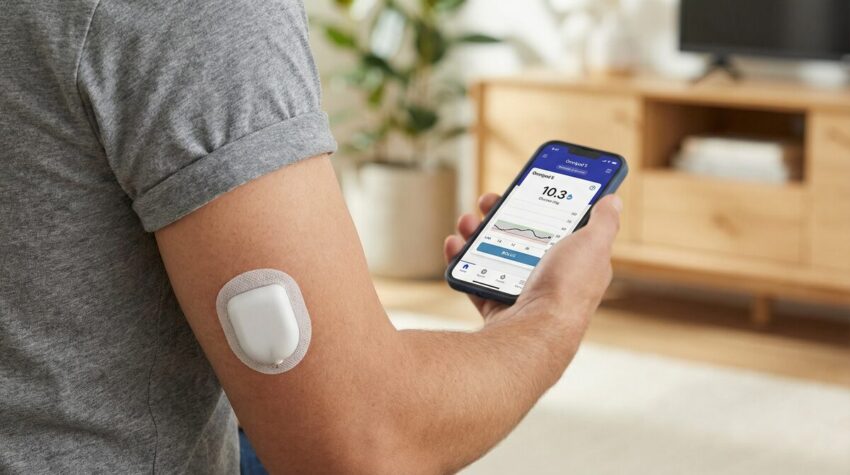
The Omnipod 5 system is fundamentally composed of three integral components: the Pod, the Omnipod 5 Application, and a compatible continuous glucose monitoring sensor.
The Pod
The Pod, a compact and waterproof** (boasting an IP28 rating, permitting submersion up to 25 feet for 60 minutes) device, is designed for direct dermal application, thereby obviating the necessity for external tubing. It incorporates an integrated insulin reservoir, capable of holding up to 200 units of U-100 rapid-acting insulin, alongside an advanced automated insulin delivery algorithm. The cannula insertion process is entirely automated and largely imperceptible, rendering it an attractive option for individuals who experience needle phobia. Its unobtrusive design facilitates comfortable wear beneath conventional attire.
The Omnipod 5 Application
The Omnipod 5 Application, accessible via a compatible smartphone† or a dedicated Controller provided by Insulet, functions as the central command interface. It encompasses a SmartBolus Calculator, an Activity feature, and Custom Foods functionalities, furnishing users with a comprehensive suite of tools for meticulous insulin management. Furthermore, the application furnishes real-time glucose and insulin data, thereby empowering users with informed decision-making capabilities.
Automated Insulin Delivery Algorithm
The intrinsic intelligence of the Omnipod 5 resides within its proprietary algorithm, which is seamlessly integrated into the Pod itself. This algorithm maintains continuous communication with a compatible CGM sensor (including Dexcom G6, Dexcom G7, and FreeStyle Libre 2 Plus Sensor) to forecast future glucose levels up to 60 minutes in advance. Leveraging these predictive analytics and a user-customizable target glucose range (typically spanning 110-150 mg/dL), the system autonomously augments, diminishes, or suspends insulin delivery at five-minute intervals. This proactive methodology is designed to sustain glucose levels within the desired therapeutic window, thereby minimizing glycemic excursions.
ℹ️ Integral Role of CGM Integration
The Omnipod 5 system is meticulously engineered to achieve optimal performance when synergistically integrated with a continuous glucose monitor. The CGM furnishes real-time glucose data, which is indispensable for the system’s automated insulin delivery algorithm to execute precise and timely adjustments. In the absence of CGM integration, the system operates in a more rudimentary mode, necessitating a greater degree of manual intervention from the user.
Clinical Efficacy and Patient Outcomes
Rigorous clinical investigations have unequivocally demonstrated the efficacy of the Omnipod 5 in ameliorating glycemic control across diverse demographic cohorts. Research consistently indicates substantial reductions in glycated hemoglobin (HbA1c) levels and an augmented time in range (TIR), concurrently diminishing the incidence of hypoglycemic episodes. For instance, a seminal study involving individuals with type 1 diabetes, aged between 6 and 70 years, reported significant improvements in the average duration of both hyperglycemic and hypoglycemic states, alongside a notable reduction in mean HbA1c from 7.16% to 6.78% in adult and adolescent participants, and from 7.67% to 6.99% in pediatric subjects, following three months of Omnipod 5 utilization [2]. Commensurate positive outcomes have been documented in very young children (aged 2-5.9 years) and adults afflicted with type 2 diabetes [3, 4].
Salient Advantages of the Omnipod 5
- Tubeless Architecture: This design confers unparalleled liberty and discretion, effectively eliminating the encumbrance of tubing and mitigating the propensity for tangles or inadvertent disconnections.
- Automated Insulin Delivery: This feature substantially mitigates the cognitive burden associated with diabetes management by automating basal insulin adjustments and facilitating proactive glucose correction.
- Waterproof Pod: The Pod’s waterproof characteristic enables participation in aquatic activities without the imperative for system disconnection.
- Enhanced Usability: The intuitive application interface and the automated cannula insertion mechanism simplify the user experience, rendering it an amenable option for novice pump users or those predisposed to needle phobia.
- Improved Glycemic Control: Empirical clinical data substantiates its effectiveness in reducing HbA1c and augmenting TIR, thereby contributing to superior long-term health trajectories.
⚠️ Crucial Considerations for Prospective Users
While the Omnipod 5 confers a multitude of advantages, its suitability may not be universal. Individuals necessitating exceptionally minute basal insulin dosages might encounter less precise dose accuracy. Furthermore, the discernible profile of the Pod could present challenges for secure site placement during strenuous physical activities or contact sports. Moreover, users frequently exposed to magnetic resonance imaging (MRI) or analogous technologies will be required to replace the entire Pod if metallic components necessitate removal. Individuals exhibiting highly labile basal insulin requirements or those who prefer extensive manual oversight of their insulin delivery regimen may perceive the automated system as less adaptable than desired. A thorough discourse regarding these factors with a qualified healthcare provider is paramount.
Potential Limitations and Practical Considerations
Notwithstanding its numerous benefits, the Omnipod 5 is not devoid of certain limitations. The system’s precision in dose delivery can be suboptimal for individuals requiring exceedingly small basal insulin increments. The physical dimensions of the Pod may also impede optimal site placement during high-impact physical endeavors. Additionally, for users undergoing frequent MRI scans or similar diagnostic procedures, the necessity to remove metallic components mandates the replacement of the entire Pod. Patients with highly fluctuating basal insulin needs or those who prefer a more granular, active role in their insulin management may find the automated paradigm less congruent with their preferences.
Ideal Candidates for Omnipod 5 Therapy
- Individuals who seek a streamlined, less intrusive methodology for diabetes management.
- Those who harbor needle phobia and are apprehensive about conventional insulin pump insertion techniques.
- Physically active individuals, including swimmers, who derive benefit from a waterproof and tubeless delivery system.
- Patients aiming to optimize their glycemic control and diminish the psychological burden associated with diabetes.
- Individuals diagnosed with type 1 diabetes, aged two years and above, and those with type 2 diabetes, aged eighteen years and older.
Concluding Remarks
The Omnipod 5 Automated Insulin Delivery System unequivocally signifies a monumental stride in diabetes technology. It offers a distinctive amalgamation of tubeless convenience, intelligent automation, and demonstrable improvements in glycemic outcomes. While it undeniably presents a compelling therapeutic option for a substantial cohort of patients, a meticulous understanding of its inherent advantages and potential limitations is indispensable for judicious decision-making. As is customary with any medical device, an in-depth consultation with a qualified healthcare professional is imperative to ascertain whether the Omnipod 5 harmonizes with an individual’s specific requirements, lifestyle, and overarching treatment objectives.
✅ Key Takeaway
The Omnipod 5 represents a tubeless, automated insulin delivery system that significantly simplifies diabetes management and enhances glycemic control for a broad spectrum of users. Its waterproof Pod and intuitive application interface afford users considerable freedom and ease of use. Nevertheless, critical considerations pertaining to basal dose accuracy, engagement in physical activities, and individual preferences for management should be thoroughly deliberated with a healthcare provider to ensure optimal therapeutic alignment.
References
- [1] Insulet Corporation. Simplify Life with Omnipod® 5. Available at: https://www.omnipod.com/what-is-omnipod/omnipod-5
- [2] Brown S. et al. Diabetes Care (2021). Study in 240 people with T1D aged 6 – 70 years involving 2 weeks standard diabetes therapy followed by 3 months Omnipod 5 use in Automated Mode. Available at: https://www.omnipod.com/what-is-omnipod/omnipod-5
- [3] Sherr JL, et al. Diabetes Care (2022). Study in 80 people with T1D aged 2 – 5.9 yrs involving 2 weeks standard diabetes therapy followed by 3 months Omnipod 5 use in Automated Mode. Available at: https://www.omnipod.com/what-is-omnipod/omnipod-5
- [4] Pasquel FJ, et al. Presented at: ADA; June 21-24, 2024; Orlando, FL. Prospective pivotal trial in 305 participants with T2D aged 18-75 yrs. Available at: https://www.omnipod.com/what-is-omnipod/omnipod-5
Disclaimer: This article is provided for informational purposes solely and should not be construed as medical advice. It is imperative to consult with a qualified healthcare professional for any health-related concerns or prior to making any decisions pertaining to your health or treatment regimen. The information presented herein is predicated upon available research and product specifications at the time of its composition.