Understanding Insulin Resistance
Insulin resistance is a prevalent metabolic condition characterized by the impaired biological response of target tissues—primarily the liver, skeletal muscle, and adipose tissue—to insulin stimulation. This diminished sensitivity to insulin leads to a compensatory increase in pancreatic beta-cell insulin production, resulting in hyperinsulinemia. While hyperinsulinemia initially helps maintain normal blood glucose levels, it can paradoxically drive further insulin resistance, creating a vicious cycle.
The metabolic consequences of insulin resistance are far-reaching, contributing to hyperglycemia, hypertension, dyslipidemia, hyperuricemia, elevated inflammatory markers, endothelial dysfunction, and a prothrombotic state. Over time, the progression of insulin resistance can culminate in serious health conditions such as metabolic syndrome, nonalcoholic fatty liver disease (NAFLD), and type 2 diabetes (T2D).
**Pathophysiology: The Cellular Mechanism of Impaired Insulin Action**
ℹ️ Understanding Insulin Signaling
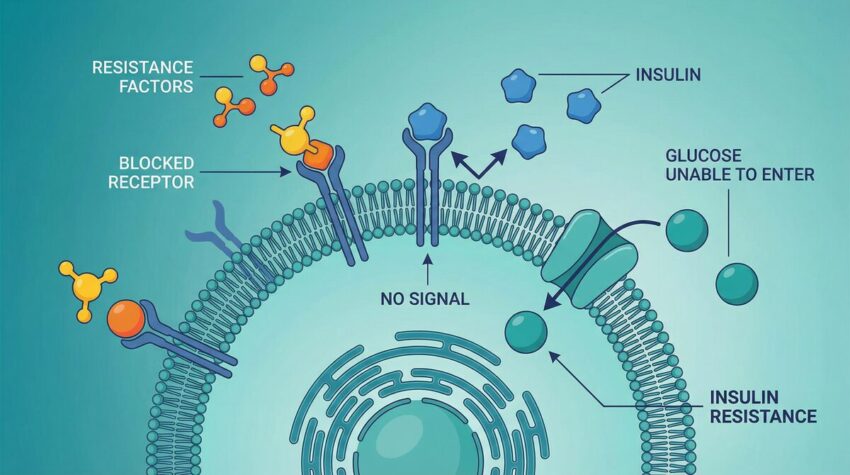
Insulin acts like a key, unlocking cells to allow glucose entry. In insulin resistance, this key doesn’t work as effectively, leading to glucose buildup in the bloodstream. This complex process involves multiple steps, from insulin binding to its receptor to the activation of intracellular signaling cascades that ultimately lead to glucose uptake.
At its core, insulin resistance involves a breakdown in the intricate signaling pathways that govern glucose uptake and metabolism. The three primary sites where this resistance manifests are:
* **Skeletal Muscle:** As the largest reservoir for circulating glucose, skeletal muscle is responsible for up to 70% of post-caloric glucose disposal. In insulin-resistant states, muscle cells accumulate intramyocellular fatty acids, particularly diacylglycerol. This molecule activates protein kinase C theta (PKC-theta), which subsequently impairs proximal insulin signaling. The consequence is reduced translocation of glucose transporter type 4 (GLUT4) to the cell membrane, leading to decreased glucose uptake by muscle tissue. The excess glucose is then shunted to the liver.
* **Hepatic Tissue (Liver):** The liver plays a crucial role in processing energy substrates, including glucose and fatty acids. When skeletal muscle becomes insulin-resistant, the liver receives an increased glucose load. Similar to muscle tissue, diacylglycerol accumulation in the liver activates protein kinase C epsilon (PKC-epsilon), impairing hepatic insulin signaling. This leads to increased *de novo* lipogenesis (DNL), where excess glucose is converted into fatty acids, contributing to hepatic steatosis (fatty liver) and ectopic lipid deposition. Furthermore, insulin’s normal suppressive effect on gluconeogenesis (glucose production) is compromised, leading to increased glucose output by the liver, exacerbating hyperglycemia.
* **Adipose Tissue (Fat):** Insulin typically suppresses lipolysis (fat breakdown) in adipose tissue. In insulin-resistant adipose tissue, particularly visceral fat, this suppression is impaired, leading to an increase in circulating free fatty acids (FFAs). These elevated FFAs can directly interfere with insulin signaling in both the liver and muscle, further intensifying insulin resistance and contributing to lipotoxicity-induced beta-cell dysfunction in the pancreas.
**Etiology: Acquired and Genetic Factors**
⚠️ Early Detection Matters
While often asymptomatic in its early stages, recognizing risk factors and subtle signs of insulin resistance is crucial for timely intervention. Early detection through regular check-ups and awareness of family history can significantly impact long-term health outcomes and prevent progression to more severe conditions.
Insulin resistance is predominantly an acquired condition, strongly linked to lifestyle factors, but genetic predispositions also play a role. Key acquired etiologies include:
* **Increased Visceral Adiposity:** Ectopic fat deposition and overflow from subcutaneous fat stores are major contributors.
* **Aging Process:** Insulin sensitivity tends to decline with age.
* **Physical Inactivity:** A sedentary lifestyle reduces skeletal muscle insulin sensitivity.
* **Nutritional Imbalance:** Diets high in calories and refined carbohydrates can stimulate excessive insulin demand.
* **Medications:** Certain drugs, such as glucocorticoids, anti-adrenergic agents, and some atypical antipsychotics, can induce insulin resistance.
* **Glucose and Lipotoxicity:** Chronic exposure to high glucose and free fatty acid levels can impair insulin signaling.
Genetic factors, while less common, can also lead to insulin resistance. These include syndromic forms like myotonic dystrophy, ataxia-telangiectasia, and lipodystrophy, as well as specific types of insulin resistance such as Type-A (due to insulin receptor gene abnormalities) and Type-B (triggered by insulin receptor autoantibodies).
**Clinical Presentation and Diagnosis**
Insulin resistance often presents subtly, with symptoms varying based on its duration, severity, and the presence of secondary complications. Common associated conditions and symptoms include:
* **Associated Diseases:** NAFLD, metabolic syndrome, prediabetes/type 2 diabetes, polycystic ovarian syndrome (PCOS), obesity, microvascular complications (retinopathy, neuropathy, nephropathy), and macrovascular complications (stroke, peripheral artery disease, coronary artery disease).
* **Associated Symptoms:** Hypertension, hyperlipidemia, increased waist circumference, acanthosis nigricans (darkening of skin folds), and menstrual irregularities/hirsutism in PCOS.
Diagnosing insulin resistance clinically can be challenging as there is no single universally accepted test. The gold standard, the hyperinsulinemic-euglycemic glucose clamp technique, is primarily a research tool due to its complexity. Clinically, insulin resistance is often inferred from the presence of metabolic syndrome, which is defined by a cluster of risk factors including elevated waist circumference, high triglycerides, low HDL, elevated blood pressure, and elevated fasting glucose.
Surrogate markers like HOMA-IR (Homeostatic Model Assessment for Insulin Resistance), QUICKI (Quantitative Insulin Sensitivity Check Index), and the triglyceride/HDL ratio are used in research and can provide clinical insights, though they are not yet integrated into routine diagnostic guidelines.
**Management and Treatment Strategies**
The cornerstone of managing insulin resistance lies in intensive lifestyle interventions:
* **Dietary Therapy:** Calorie reduction, avoidance of high glycemic index carbohydrates, and a balanced nutritional approach are crucial. This helps reduce excessive insulin demand and improve metabolic health.
* **Physical Activity:** Regular exercise enhances energy expenditure and significantly improves skeletal muscle insulin sensitivity. Studies like the Diabetes Prevention Program (DPP) have shown that lifestyle interventions, including a 7% weight loss, can reduce the onset of T2D by 58%.
While no medications are specifically FDA-approved solely for insulin resistance, several pharmacological agents are used to manage its associated conditions and improve insulin sensitivity:
* **Metformin:** Often a first-line therapy for T2D and PCOS, metformin improves insulin sensitivity and reduces hepatic glucose production. The DPP study demonstrated its effectiveness in reducing T2D onset.
* **GLP-1 Receptor Agonists (e.g., Liraglutide, Semaglutide):** These agents stimulate insulin release, inhibit glucagon secretion, and are associated with weight loss, indirectly improving insulin resistance.
* **SGLT2 Inhibitors:** By increasing urinary glucose excretion, SGLT2 inhibitors lower plasma glucose and can lead to weight loss, thereby reducing insulin resistance.
* **Thiazolidinediones (TZDs):** These drugs directly improve insulin sensitivity in muscle and adipose tissue, though their use can be limited by side effects like weight gain and fluid retention.
For qualified individuals with obesity, bariatric surgery (e.g., gastric sleeves, banding, bypass) can lead to significant fat loss and substantial improvements in insulin sensitivity, often resolving T2D.
**Conclusion**
Insulin resistance is a complex and multifaceted metabolic disorder with profound implications for public health. Understanding its underlying pathophysiology, diverse etiologies, clinical manifestations, and evidence-based management strategies is paramount for prevention and treatment. By focusing on comprehensive lifestyle modifications and, when appropriate, pharmacological interventions, individuals can effectively mitigate the risks associated with insulin resistance and improve long-term health outcomes.
✅ Key Takeaway
Insulin resistance is a foundational metabolic issue that can lead to serious health problems like type 2 diabetes. Lifestyle changes, including diet and exercise, are the most effective strategies for prevention and management. Early recognition and proactive steps are vital for maintaining long-term metabolic health.