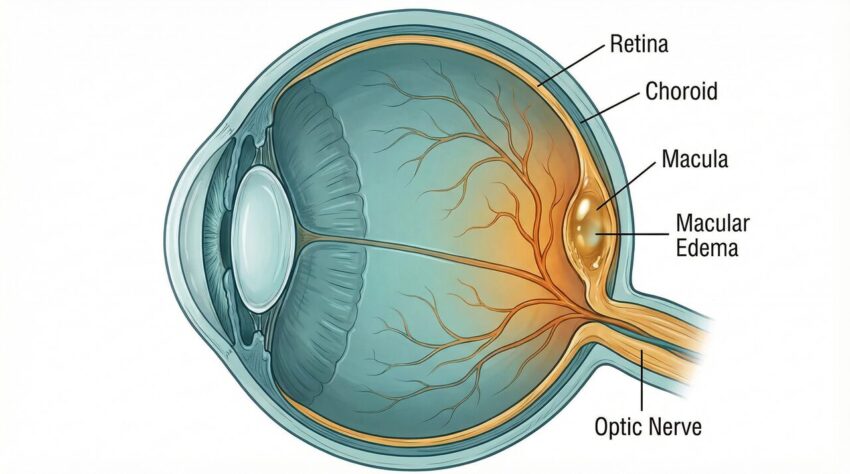
Diabetic Macular Edema (DME) is a multifaceted condition that arises primarily due to the systemic repercussions of chronic high blood sugar levels. At the heart of the issue is the disruption of the blood-retina barrier (BRB), a crucial structure that manages the exchange of substances between the bloodstream and the retina. Persistent elevated glucose levels instigate a series of harmful changes in the retinal microvasculature:
Etiology and Pathophysiology
Diabetic Macular Edema (DME) is a multifaceted condition that arises primarily due to the systemic repercussions of chronic high blood sugar levels. At the heart of the issue is the disruption of the blood-retina barrier (BRB), a crucial structure that manages the exchange of substances between the bloodstream and the retina. Persistent elevated glucose levels instigate a series of harmful changes in the retinal microvasculature:
- Endothelial Cell Dysfunction and Pericyte Loss:High blood sugar levels impair the endothelial cells, which line the retinal capillaries, and result in the loss of pericytes—cells that envelop capillaries and are vital for maintaining their integrity and barrier function. This damage weakens the tight junctions between endothelial cells, thus increasing the permeability of the blood vessels.
- Inflammation and Cytokine Release:Diabetes fosters a chronic low-grade inflammatory environment. In the retina, pigment epithelium, glial cells, and macrophages release pro-inflammatory cytokines such as interleukin (IL)-1β, IL-6, and tumor necrosis factor (TNF). These inflammatory mediators further compromise the BRB and contribute to fluid leakage.
- Vascular Endothelial Growth Factor (VEGF) Upregulation:Retinal ischemia, which occurs due to microvascular blockages and capillary loss, leads to hypoxia. This lack of oxygen stimulates the production of VEGF, a strong factor promoting both new blood vessel formation and increased vascular permeability. Elevated levels of VEGF worsen vascular leakage and can result in the formation of new, abnormal blood vessels.
- Accumulation of Advanced Glycation End Products (AGEs):Prolonged high blood sugar results in the formation and build-up of AGEs, which contribute to oxidative stress and further activate inflammatory pathways, exacerbating the breakdown of the BRB and causing fluid to leak out.
These interconnected processes lead to the accumulation of fluid, proteins, and lipids within the retinal layers, resulting in the formation of intraretinal cystic spaces and hard exudates, which are hallmarks of DME. The swelling predominantly impacts the inner nuclear layer, outer plexiform layer, Henle’s fiber layer, and subretinal space, causing distortion of the macula and diminishing visual acuity.
Diagnosis and Screening
Types of DME
DME is classified based on its proximity to the fovea, the center of the macula. Mild DME has retinal thickening and hard exudates far from the center. Moderate DME has thickening or exudates closer to, but not involving, the center. Severe DME involves the center of the macula, posing the most significant threat to vision.
Early detection and timely management are crucial in controlling DME. Given its often stealthy onset, regular eye screenings are essential for all diabetic patients. Current guidelines suggest:
- Type 1 Diabetes:Begin screening for diabetic retinopathy (DR) five years after diagnosis.
- Type 2 Diabetes:Start screening at the time of diagnosis and continue annually or more frequently, depending on the severity of systemic disease and previous findings.
The diagnostic process involves a thorough eye examination, supplemented by advanced imaging techniques:
- Optical Coherence Tomography (OCT):OCT is regarded as the gold standard for diagnosing and monitoring DME. It provides high-resolution cross-sectional images of the retina, enabling detailed visualization of retinal thickening, intraretinal fluid, subretinal fluid, and hard exudates. OCT also assists in quantifying central macular thickness (CMT) and identifying specific biomarkers that can predict treatment response.
- Fluorescein Angiography (FA):This technique involves injecting a fluorescent dye into the bloodstream and capturing rapid photographs of the retina. FA helps identify areas of vascular leakage, capillary non-perfusion, and neovascularization, offering critical insights into the extent and activity of DR and DME.
- Fundus Photography:Digital fundus photography captures images of the retina, documenting the presence and progression of DR and DME. Non-mydriatic cameras facilitate screening without the need for pupil dilation, broadening access to screening programs.
DME is commonly classified based on the severity and involvement of the fovea:
- Mild DME:Retinal thickening and hard exudates are present in the posterior pole but remain more than 1,000 μm away from the central macular subfield.
- Moderate DME:Retinal thickening or hard exudates are within the central subfield of the macula but do not involve the foveal center.
- Severe DME:Retinal thickening or hard exudates involve the foveal center, posing the greatest risk to central vision.
Management and Treatment Strategies
Managing DME requires a comprehensive approach that includes stringent control of systemic disease and targeted ocular therapies. The primary objectives are to reduce macular edema, improve visual acuity, and prevent further vision loss.
Systemic Control
Effective systemic management forms the cornerstone of DME treatment. This involves diligent control of:
- Hyperglycemia:Achieving optimal blood glucose levels (e.g., HbA1c between 6% and 7%) is crucial to slowing the progression of DR and DME.
- Hypertension:Controlling blood pressure is essential, as hypertension exacerbates microvascular damage.
- Hyperlipidemia:Managing cholesterol and lipid levels helps reduce the formation of hard exudates.
Ocular Therapies
Recent advancements have transformed DME treatment, shifting the focus from laser-based approaches to pharmacotherapy.
Anti-VEGF Therapy
Intravitreal injections of anti-VEGF agents are now the first-line treatment for DME that involves the center of the retina, especially when visual acuity is compromised. These agents work by neutralizing VEGF, thus reducing vascular permeability and inhibiting abnormal vessel growth. Commonly used anti-VEGF drugs include:
- Aflibercept (Eylea®):A fusion protein that binds to VEGF-A, VEGF-B, and placental growth factor (PlGF), providing broad inhibition of VEGF.
- Ranibizumab (Lucentis®):A recombinant humanized monoclonal antibody fragment that specifically targets and inhibits VEGF-A.
- Bevacizumab (Avastin®):An off-label, full-length monoclonal antibody that binds to VEGF-A. Although effective, its off-label use means it is not specifically approved for DME treatment by regulatory bodies.
Clinical trials, such as the DRCR.net Protocol T, have shown that aflibercept is superior in eyes with worse baseline visual acuity (20/50 or worse), while all three agents are comparably effective in eyes with better initial vision. Treatment generally involves an initial phase of monthly injections, followed by a treat-and-extend strategy tailored to each patient’s response.
Corticosteroid Therapy
Intravitreal corticosteroids are a key second-line option for patients who do not respond adequately to anti-VEGF therapy or for whom anti-VEGF agents are contraindicated. Corticosteroids reduce inflammation, stabilize the BRB, and counteract VEGF activity. Available options include:
- Dexamethasone Intravitreal Implant (Ozurdex®):A biodegradable implant that delivers sustained release of dexamethasone over several months.
- Fluocinolone Acetonide (FAc) Implant (Iluvien®):A non-biodegradable implant designed for long-term, continuous release of FAc.
While effective, corticosteroid therapy carries risks such as increased intraocular pressure and cataract formation, necessitating careful patient selection and monitoring.
Laser Photocoagulation
Historically, laser photocoagulation was the cornerstone of DME treatment. Focal or grid laser photocoagulation aims to seal leaking microaneurysms and reduce edema. With the advent of anti-VEGF therapy, laser treatment is now typically reserved for cases where anti-VEGF treatment is insufficient, or for non-center-involving DME. Subthreshold micropulse laser treatment, which minimizes retinal damage, is also utilized in specific cases.
Surgery (Vitrectomy)
Vitrectomy may be considered in select cases of DME, particularly when significant vitreomacular traction contributes to the edema. By removing the vitreous gel and relieving traction, this procedure can lead to resolution of DME and visual improvement.
Conclusion
Diabetic macular edema presents a significant challenge in diabetes management. However, advancements in diagnostic imaging and therapeutic interventions have markedly improved outcomes for many patients. A multidisciplinary approach that combines rigorous systemic disease control with personalized ocular therapies—primarily anti-VEGF injections, with corticosteroids and laser as alternatives—is crucial for preserving vision and enhancing the quality of life for those affected by this sight-threatening condition. Continued research into innovative therapies and tailored treatment strategies holds promise for further improving the prognosis of DME.
Proactive Management is Key
The cornerstone of preventing and managing DME is strict control of systemic health. Diligent management of blood sugar (HbA1c), blood pressure, and cholesterol levels can significantly slow the progression of diabetic retinopathy and reduce the risk of vision loss from macular edema.
Key Takeaway
Diabetic Macular Edema is a serious complication of diabetes, but its progression can be managed. Early detection through regular eye screenings and a multi-faceted treatment approach, including systemic health management and ocular therapies like anti-VEGF injections, are crucial for preserving vision. Advances in treatment offer hope for better outcomes.